Achievements
FATE OF PETROLEUM HYDROCARBONS AND BENZENE UNDER BIODEGRADATION IN CONTAMINATED SOILS
Ningning Sun a, Hongqi Wanga, Yongqiang Qib, Jingqi Liua, Yanjun Chena
aCollege of Water Science,
bDepartment of Geological Sciences, The University of Alabama,
Abstract:Microbial decontamination of petroleum-polluted soils has been recognized to be an efficient, economical and versatile alternative for physical or chemical treatments. In this study, the fate of different components of petroleum in contaminated soil was investigated under optimal environmental conditions which were determined by previous experiments. An aerobic bacteria species with the ability to utilize hydrocarbons and benzene as carbon and energy sources was identified as Flavobacterium sp. based on morphological, physiological and biochemical tests. It was isolated from the contaminated soil using selective enrichment culture technique. The results demonstrate a 39.88% removal rate for total petroleum hydrocarbons over a period of 27 days. Different fractions of the petroleum were observed to be degraded incongruently. The degradation rate declines with increasing alkane chain length(carbon number). It was found that the total degradation rate can be approximated by the degradation rate of the n-alkane with 23 carbon atoms. N-alkanes with carbon number lower than 23 were removed faster than the total hydrocarbon amount, and the reverse trend was observed for n-alkanes with carbon number higher than 23. Unlike n-alkanes, the degradation rate of total iso-alkanes can be simulated by that of with iso-alkanes with 19 carbon atoms. The benzene utilization studies with the bacteria species showed that it metabolizes benzene when benzene concentration is under 10mg/L, concentrations over which turn out to be inhibitory. The optimal conditions for effective removal of benzene by this species appeared to be
Key words: total petroleum hydrocarbons; n-alkanes; iso-alkanes; microorganism; benzene degradation
1 Introduction
Petroleum hydrocarbon-based fuels are one of the most prevalent soil pollutants in
Soil bio-treatment systems can be divided into two main implementation categories: in situ and above ground. In situ systems use natural environments in which additives are strategically injected in an attempt to create the necessary treatment conditions. On the other hand, use of above ground systems requires excavation of the contaminated soils followed by treatment in bioreactors. Examples of such systems include land farming (Zappi,1996;Mcginnis,1991),composting and bio-slurry treatment. The two categories mainly consist in optimizing conditions for the development of naturally occurring hydrocarbons-degraders by adding nutrients and maintaining aerobic conditions as necessary. The effects of bio-stimulation on total petroleum hydrocarbons have been extensively investigated in controlled conditions and in open field experiments under optimal conditions (Bossert,1984; Leahy,1990;Atlas,1991;Outdot,2000).
However, the major problem in studying the degradation of petroleum is its complexity because it comprises an enormous number of components. Therefore, it is necessary to study the influence factors and the ultimate fate of each fraction in optimal conditions. Moreover, several studies showed that the lighter fractions were degraded more rapidly than the heavier ones, n-alkanes were degraded more rapidly than branched alkanes and aromatics (Dutta, 2000).
In this present study, we examine the mode and fates of different components of petroleum in soil under the optimal conditions which had been determined by the former experiments. In order to investigate the biodegradation modality of specific hydrocarbon, we have also isolated one microorganism from soil to study the removal efficiency of benzene in different controlled conditions.
2 Materials and Methods
2.1 Soil and oil treatment
The soil was collected from an oil production area near Daqing oil field in Hei Longjiang province,
Two treatment systems were conducted in laboratory. One was blank group and the other was experimental. The experimental design included three replicates of each treatment. Six influencing factors were selected: oil concentration, nutrient (NO3-, PO43-), oxidant (250ml 3% H2O2 solution was added every two days), temperature (Room temperature,
Table 1 List of factorsˊ levels
Group | Concentration mg·g-1 | Nutrients mg·g-1 | Oxidant mg·g-1 | Surfactant mg·g-1 | Tempe-rature | Water ratio % |
Blank | 14 | 1.89 0.02 | 0 | 0 | Room | 3.06 |
Experiment | 34 | 1.89 0.02 | 0.015 | 1 | Room | 25 |
2.2 Petroleum hydrocarbon extraction and measurement
Soil samples were picked 5 gram in the 0,2,6,14,27 days respectively. They were extracted in 20ml tetrachloride carbon on a mechanical shaker over 12h and finally sonicated for 30min.After centrifugation ,total petroleum hydrocarbon levels in the extract were analyzed by gas chromatography with flame ionization detection (GC/FID).Using a Shimadzu GC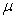 m×0.25
m×0.25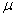 m film colum..1
m film colum..1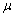 L of sample was injected and the injector and detector temperatures were 300 and 325 ℃, respectively. The temperature program was as follows: Initial temperature 100 ℃(hold 5 min), rate 4℃ /min to 300 ℃ (hold 15min) and using voyage Mass Spectrometry Detector to determine each component in the extract.
L of sample was injected and the injector and detector temperatures were 300 and 325 ℃, respectively. The temperature program was as follows: Initial temperature 100 ℃(hold 5 min), rate 4℃ /min to 300 ℃ (hold 15min) and using voyage Mass Spectrometry Detector to determine each component in the extract.
2.3 Microorganism
The microorganism adopting selective enrichment culture technique was isolated from the experimental soil mentioned above. The bacterial strain was grown on mineral salt medium containing (g/L)NH4Cl,0.1;MgCl2·6H2O,0.08;K2HPO4, 0.25; CaCl2·2H2O,0.1;distilled water 1000ml;pH,7.0;supplemented with benzene up to 1mg/L as the sole source of carbon and energy (Karigar,2006;Ganji,1992).In the metabolic studies, growth substrate(5.6 mg/L) was incorporated to the sterilized mineral salt medium. The flasks were inoculated with 5% (v/v) of inoculums.
Purified bacterium was identified and characterized based on the morphological, physiological and biochemical features as described in Bergey’s Manual of Systematic Bacteriology (Keddie,1986).
The growth curve of the organism was followed by measurement of the optical density at 650nm.All operations were performed under aseptic conditions. All the results represent data from at least three independent and repetitive experiments. The degradation of benzene was followed by ultrasonic solvent extraction with hexane and acetone from the spent medium at specific time intervals and the measurement of the optical density at 254nm.
2.4 Biodegradation of benzene in different controlled conditions
Four influencing factors were selected to investigate effects on the benzene removal. The factors were: inoculum concentration, temperature, rotary speed and pH.0%,3.0%,4.5%,5.0%,6.0%,7.0%,8.0%,9.0%,10.0%,11.0%,12.0%(v/v) of inoculums were inoculated in the flasks and incubated on rotary shaker(150rpm) at 30 ℃,pH,7.0,44 hour. Based on the result of inoculum test , the flasks were inoculated with 10% at 26℃,28℃,30℃,32℃,34℃,36℃,38℃,40℃. In order to determine the appropriate dissolved oxygen in which the benzene can reach the optimal removal efficiency, the different rotary speed was controlled. The rotary was conducted in 120 rpm,140 rpm,160 rpm,180 rpm,200 rpm,220 rpm,240rpm,10% inoculum, at 30 ℃,pH,7.0,44 hour. Use of Ca(OH)2 solution for the pH adjustment, the pH ranged from 5.7 to 7.5.
3 Results and Discussion
3.1Total petroleum hydrocarbon analysis
In this study, the initial concentration of total petroleum hydrocarbon in the soil was 34mg/L dry soil. After 27 days of experiment, total petroleum hydrocarbons were 21.79mg/L. In all treated soils, it decreased logarithmically with time. In absence of fertilization, oxidant and surfactant, the final biodegradation of total petroleum hydrocarbon reached 39.88%.Chemical analysis by GC-MS was shown in Fig.1. Compared to the left stable solvent, the chromatogram shape of petroleum hydrocarbon in sample changed flat apparently, which coincided with the decrease of total petroleum hydrocarbon. Especially, n-C23 which appeared in 47.15 minute had a sharp decline.
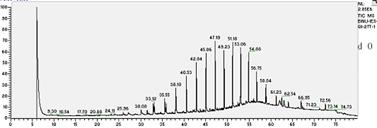

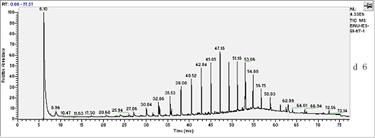
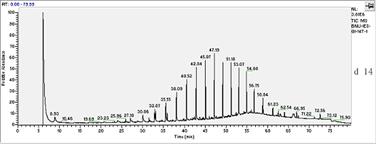
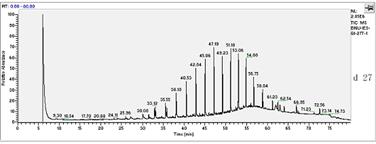
Fig.1 biodegradation of total petroleum hydrocarbon
Many studies had showed that the high molecular weight fractions derived from oil refinery sludge are exceptionally hard to remediate (Huang, 2005;Mcnicoll, 1995; Lin,2005;Buist,1997). As can be
seen from the Fig.1, the removal efficiency of petroleum components during biodegradation generally decreased with increasing alkane chain length (carbon number). For example, the concentrations in the residue were higher for alkanes with chain length greater than nC-23 as compared to those lower than nC-23.This indicated that all of the low boiling point fraction and most of the middle boiling point fraction had been removed, but most of the high boiling point fraction was present in the residues .
In the present study, we believe that the concentration increase of long chain alkanes in degradation most likely resulted from a combination of their lower removal efficiency and a decrease in oil volume after the degradation. Moreover, higher concentration reduction of the total petroleum hydrocarbon for the petroleum most likely resulted from a greater abundance of the short chain alkanes in the oil. In addition, from day 0 to day 27 experiment, the baseline of chromatogram was driven up gradually. This result may be caused by branch-alkanes. With the large amounts of hydrocarbons were degraded, the removal efficiency of branch-alkanes were lower than that of the n-alkanes, so the relative abundance of branch-alkanes was higher. Moreover, it was possible that much new branch-alkanes were produced and appeared simultaneity, so that raised the baseline.
3.2 Oil Composition analysis
Under the optimal controlled conditions, total amount of the n-alkanes decreased from 2.25 gram per 100 gram soil at 0 day to 1.46 gram at 27 day. The degradation rate to shortest-chain alkane was highest and the efficiency decreased with increasing C numbers. However, the relative abundance of alkanes increased with increasing chain length, plateaued between n C-23 to nC-29, as shown in Fig.2.The degradation rate of n C-16 and n C-19 at the initial period was higher than that of later phase. The removal rate of low molecular weight fractions was greater than that of high ones. The relative amount of nC-23 changed 0.41% in the 27days. This indicated that the rate of n C-23 was close to removal rate of the total hydrocarbons and n-alkanes with C lower than 23 were removed more quickly than that of total hydrocarbons.
Iso-alkanes had the similar mode with the n-alkanes(Fig.3). With the increase of molecular weight, the degradation rate of iso-alkanes decreased gradually. However, unlike n-alkanes, the degradation of iso-alkane with C number more than 19 alkanes was lower than that of the total hydrocarbons, while the rate of n C-23 was close to the total ones. This might suggest that the degradation of iso-alkanes were lower than that of n-alkanes.
The change trend of cyclo-alkanes was shown in Fig.4. In total, the degradation rate of cyclo-alkanes was higher than that of total petroleum hydrocarbons. However, when the C number was much higher, the influence of ring became less, so the degradation rate would decrease.
3.3 Microorganism
Microorganisms have the ability to adapt to a variety of environmental conditions. In the present work, the preliminary objective was to isolate a microorganism that could grow on benzene. A microorganism was obtained that could metabolize benzene up to 10mg/L, by adopting the enrichment culture technique .The classification of bacteria was carried out entirely based on phenotypic characteristics, such as cell wall type, morphology, motility and nutritional requirement. The isolated benzene metabolizing strain was thus subjected to various morphological, physiological and biochemical tests. The results of the tests showed that the strains growing on benzene was Gram negative, without flagellum, rod, unmovable and aerobic. These results showed that the strain degrading benzene belonged to Flavobacterium sp. according to Bergey’s Manual of Determinative Bacteriology (Keddie, 1986).
The organism tolerated benzene concentration up to 10mg/L, but at higher concentrations there was a decline in the growth due to substrate inhibition. The study demonstrated that the bacterial strain could adapt to degrade the contaminants if given sufficient time. Acquisition of degrading abilities by selective enrichment has been in laboratory ecosystems for many organic compounds and heavy metals (Alexandaer, 1999; Vander, 1992).
3.4 The growth of strain
As shown in Fig.5, from inoculation to 4 hour, the period belonged to lag phase and microorganism began to adapt to the new growth enrichment, the physiological phase came back. In this period, it was just the growth of individual cell rather than the increase of total amount. From 4h to 20h, this was accelerated period, with the growth of individual cells, the initial cell division began, and the number of cell increased. From 20h to 32h, this was exponential phase. During this period, the assimilation was greater than that of metabolic. The material demand from the environment of microorganisms was much more. From 30h to 36h, the cell was decelerated phase. Cells did not grow after division. At this time, the demand of Carbon source and nutrients decreased sharply. From 36h to 44h, cell was stationary phase and assimilation counterpoised with the metabolism. After 44h, cell was contabescence phase, in which the overall number of microorganism was no longer increasing.
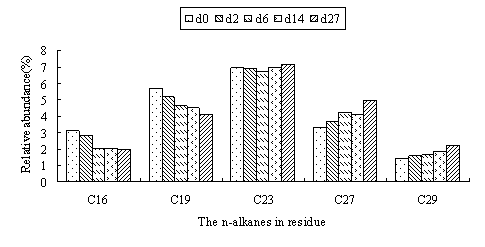

Fig.2 The relative abundance of n-alkanes in residue
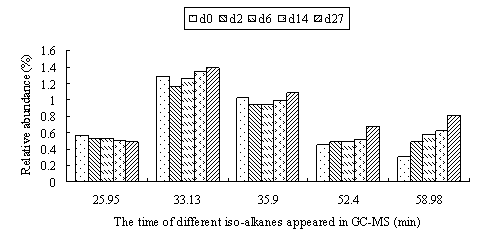
Fig.3 The relative abundance of different iso-alkanes in residue
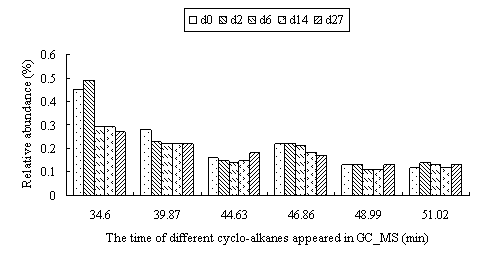
Fig.4 The relative abundance of different cyclo-alkanes in residue

Fig.5 The growth of strain during 44 hour incubation
3.5Effects of different controlled conditions
3.5.1 Inoculum concentration
As can be seen from the Fig.6a, with the increase of inoculum, the efficiency of benzene degradation increased correspondingly. Especially the concentration ranged from 2.0% to 10%, the rate of biodegradation rose obviously. However, when the concentration exceeded 10%, the degradation declined. It has been reported that bacterial concentration did not
limit the biodegradation potential of a given population and that no significant correlation was noticed between the concentration of oil- degradative bacteria and hydrocarbon biodegradation potentials (Kim, 2005; Venkateswaran,1991;Venosa,1992).In our study, however, even though low concentrations of inoculum were not significantly effectively, inoculation using higher concentrations markedly enhanced benzene degradation. These results suggest the importance of high concentrations of microorganisms for enhancing the mineralization of benzene; despite the practical conditions involved in the preparation of large amounts of microorganisms for seeding and competition with the natural microflorm under changing environmental conditions.
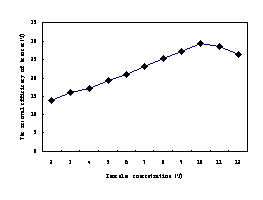
Fig.6a The removal efficiency of benzene influenced by inoculum concentration
3.5.2 Temperature
The result of temperature influencing the benzene biodegradation was shown in Fig.6b.When the temperature was lower than 30℃, the efficiency of degradation with the temperature heightened. The most appropriate temperature was 30℃ and the degradation was 34.5% at this temperature. In contrast, when the temperature was greater than 30℃, the capability of degradation for the organism markedly declined. In particular at 40℃, the efficiency of removal was only 11.3%,which was third of that at 30℃.Among the other parameters controlling hydrocarbon biodegradation, temperature is generally considered as one of the most important, since degradation is thought to obey to the Arrhenius relationship (Delillea,2004).Microbial metabolism increases as temperature increases (Leahy,1990),usually doubling for each 10℃ increase in temperature from 10 to 40℃(Bossert,1984).However, from the results in this study, the optimal temperature for the degrading benzene was 30℃.
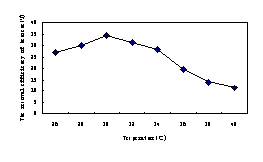
Fig.6b The removal efficiency of benzene influenced by Temperature
3.5.3 The rotary speed
The aerobic microorganisms, the dissolved oxygen was also considered one of the most important factors influencing microbial metabolism and degradation behavior. In order to determine the appropriate dissolved oxygen in which the benzene can reach the optimal removal efficiency, the different rotary speed was controlled .From the Fig.6c, at the same other conditions, the optimal rotary speed was 180-200rpm for benzene degradation. This may suggest that with the rotary speed increase, the concentration of oxygen increased correspondingly in the system. Moreover, the increased rotary speed might intensify the contact probability between the bacteria and benzene. But over-speed might shorten the contact time and decrease the efficiency of removal.
Fig.6c The removal efficiency of benzene influenced by rotary speed
3.5.4 The pH
The pH of culture solution was adjusted from 5.7 to 7.5 to examine the potential influence of pH on the microbial activity. Use of Ca(OH)2 solution for the pH adjustment was presumed to have minimal effect on the microbial activities(Zhang,2005;Kastner,1998).The optimal pH values reported in the literature for the biodegradation of hydrocarbons ranged between 6.5 and 8.0 (Morgan,1989).In this study, as shown in Fig.6d, we observed the high degradation rate with the pH between 6.5 and 6.7, and greater or lower the value, the efficiency of removal decreased.
![]()
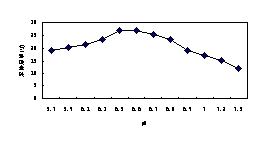
Fig.6d The removal efficiency of benzene influenced by pH
4 Conclusions
The present investigation supported the proposition that in optimal controlled conditions the number of C affected the degradation of each fraction of oil, the more C, the more difficult for degradation, and with the same C, the removal efficiency depended on the functional group. N-alkanes with C lower than 23 were removed more quickly than that of total hydrocarbons, while the ones greater than 23 were slower than that. A microorganism recognized as Flavobacterium sp. was isolated from contaminated soil which could metabolize benzene up to 10mg/L. Through a series experiment of single factor influencing degradation, the result showed that the inoculum concentration, temperature, initial pH and shaker speed all affected the efficiency of benzene removal. The optimal condition was 10% bacteria inoculated in the cultured solution with pH 6.5-6.7,30 ℃,shaker speed 180-200rpm.
Acknowledgement
This work was supported by Nature Science Foundation of China (40472129).
References
[1] Alexander M. Biodegradation and Bioremediation. 1992, Academic Press, San Diego, CA.
[2] Atlas, R.M.. Microbial hydrocarbon degradation—bioremediation of oil spills. Journal of Chemistry Technology and Biotechnology . 1991, 52,149–156.
[3] Bossert, I., Bartha, R.. The fate of petroleum in soil ecosystem, in: Atlas, R.M. (Ed.), Petroleum Microbiology. Macmillan Co., New York. 1984, 435–476.
[4] Buist, I.; Trudel, K.; Morrison, J.; Aurand, D. Laboratory studies of the properties of in-situ burn residues. Proceedings of the 1997 Oil Spill Conference; American Petroleum Institute: Washington, DC, 1997, 149-156.
[5] C.H.Chaineau;G..Rovgewx; C.Yepremian; J.Oudot. Effects of nutrient concentration on the biodegradation of crude oil and associated microbial populations in the soil. Soil Biology & Biochemistry. 2005, 37, 1490-1497.
[6] Chaineau.C.H; Morel.J.L;Outdot,J. Biodegradation of fuel oil hydrocarbons in the rhizosphere of Maize(zea mays L.).Journal of Environmental Quality.2000, 29, 569-578.
[7] Chaineau.C.H;Morel.J.L;Outdot,J. Microbial degradation in soil microcosms of fuel oil hydrocarbons from drilling cuttings. Environmental Science and Technology 1995, 29, 1615-1621.
[8] [Delillea. Daniel; Coulona. Frederic; Pelletier Emilien. Effects of temperature warming during a bioremediation study of natural and nutrient-amended hydrocarbon- contaminated sub-Antarctic soils. Cold Regions Science and Technology. 2004, 40, 61– 70.
[9] Dutta.T.k;Harayama shigeaki. Fate of crude oil by the combination of photooxidation and biodegradation . Environmental Science and Technology.2004, 34, 1500-1505.
[10] Ganji SH; Pujar BG.. Bacterial degradation of Benzalphthalide. World J.Microbial. Biotechnol.. 1992,8: 324
[11] [Huang.X.D; Alawi.Y.E ; Gurska. Jolanta,;Bernard R. Glick, Bruce M. GreenbergT.A multi-process phytoremediation system for decontamination of persistent total petroleum hydrocarbons (TPHs) from soils Microchemical Journal. 2005, 81,139– 147.
[12] Karigar.C;Mahesh.A;Nagenahali,M;Yun,D.J. Phenol degradation by immobilized cells of Arthrobacter citreus.Biodegradation.2006,17,47-55.
[13] Kastner, M.; Breuer-jammali, M.; Mahro, B. Impact of inoculation protocols, salinity, and pH on the degradation of polycyclic aromatic hydrocarbons (PAHs) and survival of PAH-degrading bacteria introduced into soil. Appl. Environ. Microbiol. 1998, 64, 359-362.
[14] Keddie RM, Collins MD, Jones D Genus Arthrobacter. In: Bergey’s Manual of Systematic Bacteriology, Williams and Wilkins, Baltimore. 1986,Vol 2 ,1288–1300.
[15] Kim Sang-Jin, Dong Hyuk Choi, Doo SuepSim. Evaluation of bioremediation effectiveness on crude oil-contaminated sand .Chemosphere ,2005, 59, 845–852.
[16] Leahy, S.G., Colwell, R.R.. Microbial degradation of hydrocarbons in the environment. Microbiology Research . 1990, 54, 305–315.
[17] Lin.Q.X.;Irving A;Mendelssohn.K.C. In-Situ Burning of Oil in Coastal Marshes. 2. Oil Spill Cleanup Efficiency as a Function of Oil Type, Marsh Type, and Water Depth. Environmental Science and Technology.2005, 39,1855-1860.
[18] Mcginnis.G.D;Borazjani.H;Pope.D.F;Strobel.D.A and Mcfarland.L.K. On-site treatment of creostote and pentachlorophenol sludges and contaminated soil.Robert S.kerr Environmental Research Laboratory Office of Research and Development. US Environmental Protection Agency,Ada,Oklahoma.1991.
[19] McNicol. D.M.; Baweja. A.S. Bioremediation of petroleum-contaminated soils: an innovative, environmental friendly technology, Environment Canada, 1995.
[20] Morgan, P.; Watkinson, R. J. Hydrocarbon degradation in soil and methods for soil biotreatment. Biotechnol. 1989, 8, 305-333.
[21] Oudot, J.. Biodegradabilite´ du fuel de l’Erika. Comptes Rendus de l’Academie des Sciences III-Vie.2000, 323, 945–950.
[22] Qi Yongqiang;Wang.H.Q.;Liu.J.Q. Factors affecting the microbial degradation of soil oil orthogonal experimental analysis. Acta Geoscien Tia Sinica.2003, 24,279-284
[23] Suguira,k.;Ishihara,M.;Shimauchi,T.;Harayama,S. Physicochemical properties and biodegradability of crude oil. Environmental Science and Technology . 1997, 31, 45-51.
[24] vander Meer JR; de Vos WM, Harayama S ; Zehnder AJB. Molecular mechanisms of genetic adaptation to xenobiotic compounds. Microbiol. Rev. 1992, 56, 677–694.
[25] Venkateswaran, K., Iwabuchi, T., Matsui, Y., Toki, H.,Hamada, E., Tanaka, H. Distribution and biodegradation potential of oil-degrading bacteria in North Eastern Japanese costal waters. FEMS Microbial. Ecol. 1991, 86, 113–122.
[26] Venosa, A., Haines, J.R., Nisamaneepong, W., Govind, R.,Pradhan, S., Siddique, B. Efficacy of commercial products in enhancing oil biodegradation in closed laboratory reactors. J. Ind. Microbial. 1992,10, 13–23.
[27] Zappi .M E.;Rogers,B.A;Teeter.C.L;Gunnison.D;Bajpai.R. Bioslurry treatment of a soil contaminated with low concentration of total petroleum hydrocarbons. Journal of Hazardous Materials.1996, 46,1-12.
[28] Zhang.P;Sheng G Y;Feng Y C;Miller D M. Role of Wheat-Residue-Derived Char in the Biodegradation of Benzonitrile in Soil: Nutritional Stimulation versus Adsorptive Inhibition. Environmental Science and Technolgy..2005, 39, 5442-5448.




